Atomic number and atomic mass
|
Argon has the atomic number of 18 and and atomic mass of 40.
|
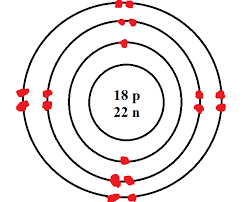
Bohr diagram |
Protons,Electrons,and Neutrons |
|
18 electrons so 8 are on the valance and the element is stable.
A.P.E M.A.NAtomic Number is equal to Number of Protons and Electrons.
To get your Neutrons you need to use Mass number-Atomic number=Neutrons.40-18=22 |
Protons and electrons are 18,and neutrons 22 after you do M-A=N.
Group and period,periodic table and elementPeriod 3 and group 18.its a noble gas and a non-metal.
|
FUN FACTS
|
Argon makes 1% of the atmosphere.
It is used to fill incasadent and fluorescent light bulbs. Argon is used for welding,cutting,and spraying metals. |
Boiling point 185.85 degrees C.Melting point 189.35 degrees C.
Argon cost $2.80 or $4.80 argon comes from the Greek word "argos" meaning "lazy" or "inactive." |